Authors: M. Gambacciani, M. Levancini and M. Cervigni
Published in Climacteric. 2015;18(5):757-763
1. HIGH IMPACT JOURNAL
Published in Climacteric, the Journal of the International Menopause Society (IMS). The journal was founded in 1998 and has become a leader in publishing peer-reviewed research on menopause.
2. HORMONE-FREE TREATMENT FOR THE SYMPTOMS OF GENITOURINARY SYNDROME OF MENOPAUSE (GSM)
GSM (or vulvovaginal atrophy) is a chronic condition, which affects up to half of all postmenopausal women. Symptoms of GSM include dryness, burning, irritation, lack of lubrication and impaired sexual function. Traditional treatments have been limited to local or systemic estrogen therapy.
3. MATERIALS AND METHODS
45 postmenopausal women with symptoms of GSM were treated with a non-ablative vaginal erbium laser (Fotona, Slovenia). As a control group, 25 postmenopausal women were treated with an established treatment for GSM (1g of vaginal gel containing 50 µg of estriol twice weekly, for 3 months).
4. SIGNIFICANT IMPROVEMENT OF VAGINAL DRYNESS AND DYSPAREUNIA
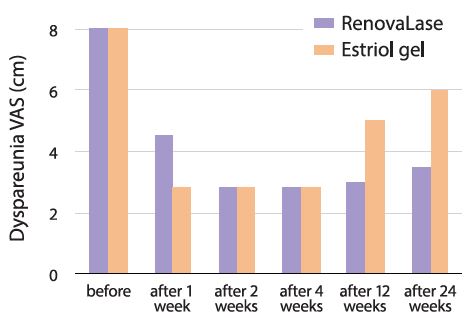
RenovaLase treatment resulted in a rapid and long-lasting improvement in the signs and symptoms of GSM. This treatment is of special importance in the treatment of postmenopausal women who cannot be treated with hormones.
Effect of RenovaLase therapy on dyspareunia using the visual analog score (VAS) on a 10-point scale for the women receiving laser treatment and the women receiving estriol. In the estriol group, a reduction of efficacy can be seen 12 weeks after the end of treatment. Conversely, the RenovaLase group maintained the same positive results throughout the entire study period up to the 6-month follow-up.